MHRA issues recall for blood pressure medication after packaging mix-up

Pharmacies and healthcare professionals have been urged to return the remaining stock to suppliers
Don't Miss
Most Read
Latest
The Medicines and Healthcare products Regulatory Agency has issued a recall for a single batch of Ramipril 5 mg Capsules manufactured by Crescent Pharma Limited.
The precautionary measure concerns batch number GR164099, following concerns that a manufacturing fault may have caused two blood pressure treatments to be incorrectly packaged.
Specifically, blister strips containing Amlodipine 5 mg Tablets may have been placed inside cartons labelled as Ramipril capsules.
Those prescribed Ramipril are being urged to examine their medication packaging for the affected batch number and return any packs containing blister strips marked "Amlodipine" to their local pharmacy.
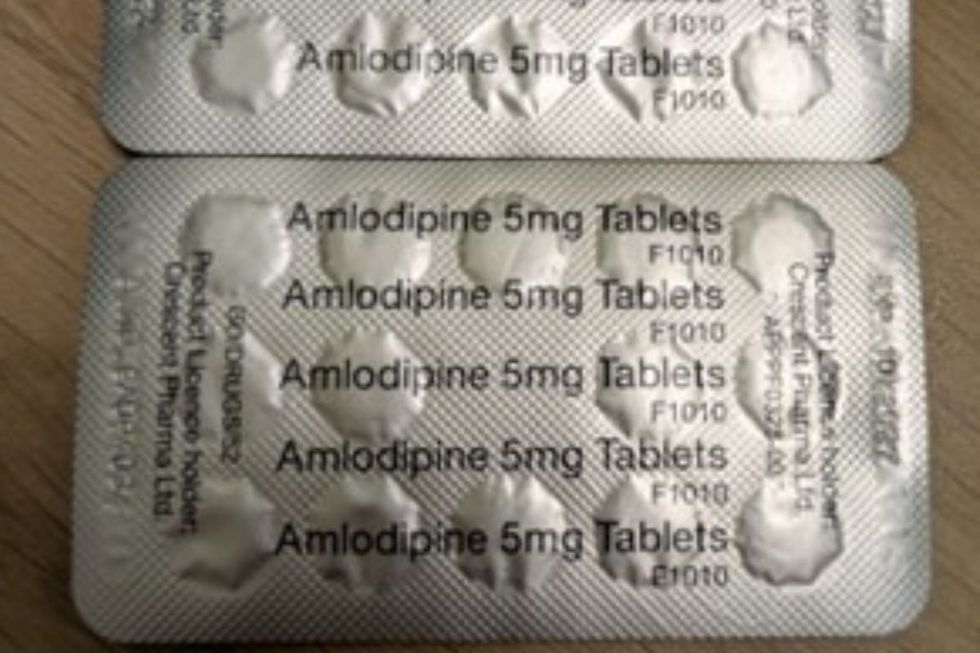
The recall concerns blister strips containing Amlodipine 5 mg Tablets
|GOV.UK
The packaging error came to light after a pharmacy lodged a complaint when a patient discovered their sealed Ramipril carton contained Amlodipine blister strips instead.
Both blood pressure medicines are manufactured by Crescent Pharma at the same production facility.
The mix-up appears to have occurred during the secondary packaging stage, when blister strips are placed into their outer cartons.
Ramipril capsules are described as light grey and green gelatine capsules marked with "R" on the cap and "5" on the body.
By contrast, Amlodipine tablets are white to off-white, round tablets with "5" debossed on one side.
Shareen Doak, Deputy Director of Benefit-Risk Evaluation at the MHRA, said: "If you take Ramipril, check the packaging for batch number GR164099.
"The batch number and expiry date information can be found on the outer carton. If you have received this batch, check that the medication name on the carton matches the blister strips inside."
She added: "If the carton contains blister strips that are labelled as Amlodipine 5 mg tablets, contact your dispensing pharmacy.
"If the carton contains blister strips that are correctly labelled as Ramipril 5 mg Capsules, you do not need to take further action."
LATEST DEVELOPMENTS
The regulator has emphasised that the health risk from this accidental substitution remains low, with dizziness from reduced blood pressure being the most probable adverse effect.
Patients who believe they may have inadvertently taken the Amlodipine tablets and are experiencing side effects should seek immediate medical attention.
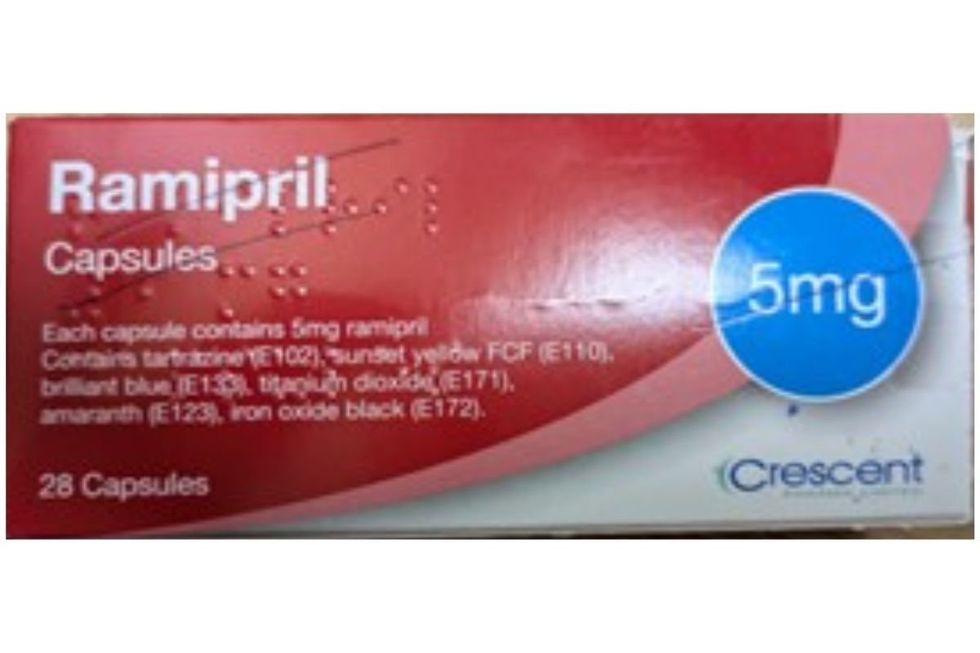
Those prescribed Ramipril are being urged to examine their medication packaging
|GOV.UK
The MHRA advises bringing the medication leaflet and any remaining tablets when visiting a pharmacy or GP surgery.
Ms Doak offered reassurance: "If you've already taken Amlodipine, please be reassured that there is a very low risk to your health."
Since both medicines treat high blood pressure, the primary concern is that an unfamiliar medication may cause blood pressure to drop lower than usual, potentially resulting in dizziness.
Any suspected adverse reactions should be reported through the MHRA Yellow Card scheme.
Our Standards: The GB News Editorial Charter