'Check your pills!' Urgent warning over antidepressant mix-up linked to nausea and headaches

The recall comes after a patient noticed the wrong antidepressant medication had been placed inside its packaging
Don't Miss
Most Read
The Medicines and Healthcare products Regulatory Agency has issued a precautionary recall affecting a single batch of Sertraline 100mg tablets after discovering the wrong antidepressant medication had been placed inside some packaging.
Amarox Limited, the manufacturer, is withdrawing batch number V2500425 of its film-coated sertraline tablets following the identification of a packaging mix-up.
The error came to light after a patient reported finding Citalopram 40mg tablets inside their sealed sertraline carton.
Both medications belong to the SSRI class of antidepressants, which function by increasing serotonin levels in the brain to treat depression and anxiety disorders.
TRENDING
Stories
Videos
Your Say

Patients taking sertraline have been urged to inspect their medication immediately
|GETTY
Patients prescribed sertraline who possess this particular batch have been urged to inspect their medication immediately.
It appears the manufacturing fault took place during the secondary packaging process, when blister strips are placed into their outer cartons.
Since Amarox Limited produces both sertraline and citalopram at the same production site, the mix-up resulted in a citalopram blister strip ending up inside a sertraline box.
The MHRA has classified this as a Class 2 recall, prompting swift action from the regulator to protect public safety.
Anyone who suspects they may have inadvertently consumed the incorrectly packaged citalopram tablets, or who is experiencing any adverse effects, should seek medical guidance without delay.
Dr Alison Cave, the MHRA's Chief Safety Officer, has provided clear instructions for those who may be affected.
LATEST DEVELOPMENTS
"If you have been prescribed Sertraline 100mg tablets and have received batch number V2500425, please check that the carton contains the right medication. You can find the batch number and expiry date printed on the side of the outer packaging," she said.
"If the blister strips inside the carton are labelled Citalopram 40mg, please contact your pharmacy as soon as possible. If they are labelled "Sertraline 100mg", no further action is needed."
Dr Cave noted individuals who have mistakenly taken citalopram alongside or instead of their prescribed sertraline may notice heightened serotonergic effects, including nausea, headaches, disrupted sleep patterns, and mild anxiety.
Healthcare professionals have been directed to cease supplying the affected batch immediately and return all remaining stock to their suppliers.
Pharmacists and other dispensing staff must identify patients who may have received the compromised product and request its return if any medication remains.
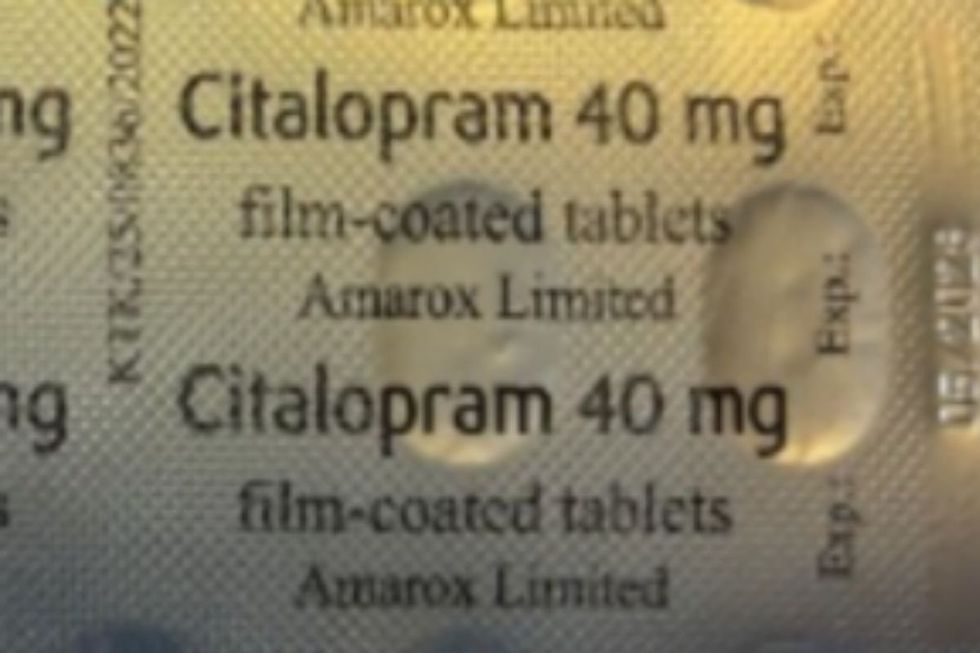
Citalopram 40mg tablets have been discovered inside sealed sertraline cartons
|GETTY
Where affected patients are identified, healthcare professionals should liaise with the patient's GP or responsible clinician to arrange a treatment review and determine whether a fresh prescription is necessary.
Certain patient groups warrant particular attention and monitoring, including those aged over 65 or under 18, individuals with cardiac or liver conditions, and those whose bodies metabolise medications differently.
Any suspected adverse reactions should be reported through the MHRA Yellow Card scheme.
Our Standards: The GB News Editorial Charter










